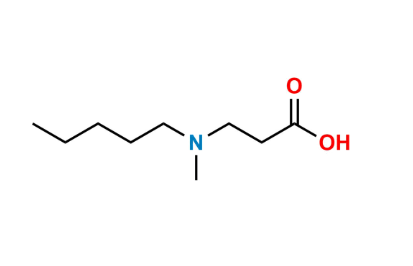
Reference Standard
| CAS Number | 625120-81-2 |
| Formula | C₉H₁₉NO₂·HCl |
| Molecular Weight | ~209.71 g/mol |
| Purity | ≥95.0% |
| Storage | Store at 2–8 °C, dry and well-sealed |
A European Pharmacopoeia–listed related compound formed during ibandronate synthesis, representing an intermediate or side-chain degradation product.
Used as an official EP impurity reference for method validation, system suitability testing, and regulatory impurity quantification.
HPLC/UPLC impurity profiling, LC-MS identity confirmation, and reference retention time matching.