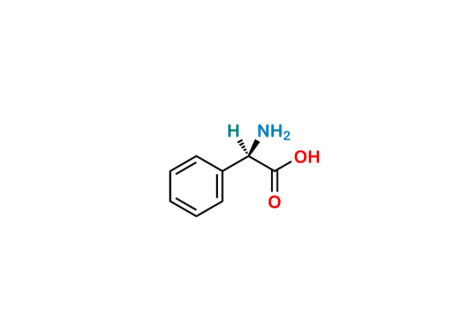
Reference Standard
| CAS Number | Not Assigned |
| Formula | C₁₉H₂₃N₃O₆S |
| Molecular Weight | ~425.46 g/mol |
| Purity | ≥ 95.0% |
| Storage | Store at 2–8°C, protect from moisture |
Bacampicillin EP Impurity B is a specified impurity listed in the European Pharmacopoeia, used for impurity profiling and compliance testing of Bacampicillin.
EP monograph compliance
Quality control testing
Analytical method validation
HPLC (EP Method)
LC-MS
IR Spectroscopy