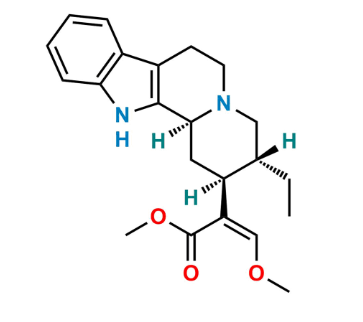
Reference Standard
| CAS Number | Not assigned |
| Formula | As defined in the EP monograph |
| Molecular Weight | Approximately 350–365 g/mol |
| Purity | As per EP CRS specification |
| Storage | Store refrigerated; protect from light and humidity |
EP Impurity E is a defined related substance of yohimbine monitored for pharmacopoeial compliance and stability control.
Used as an EP reference standard for impurity limit testing, stability-indicating method validation, and regulatory submissions.
Analyzed by EP-specified HPLC/UPLC methods, with LC-MS and spectral data supporting identification.