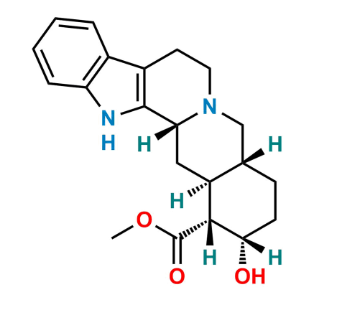
Reference Standard
| CAS Number | Not assigned |
| Formula | As per European Pharmacopoeia definition |
| Molecular Weight | Approximately 335–360 g/mol |
| Purity | EP reference standard grade |
| Storage | Store at 2–8 °C; keep container tightly closed |
Yohimbine EP Impurity D is a specified EP impurity formed through stereochemical or oxidative transformation of yohimbine.
Used in EP-compliant impurity testing, stability studies, and quality control of yohimbine API.
HPLC impurity profiling per EP, LC-MS confirmation, and system suitability testing.