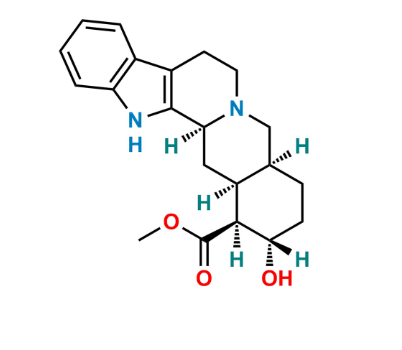
Reference Standard
| CAS Number | Not assigned |
| Formula | As defined in the EP monograph |
| Molecular Weight | Approximately 350 g/mol |
| Purity | As per EP CRS requirements |
| Storage | Refrigerated (2–8 °C); protect from light |
EP Impurity B is a pharmacopoeially specified yohimbine-related compound, representing a structural modification of the parent molecule.
Used for regulatory impurity profiling, analytical method validation, and batch release testing of yohimbine.
Determined by HPLC/UPLC according to EP, supported by LC-MS and spectral comparison.