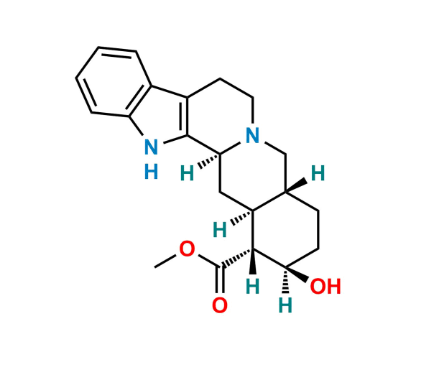
Reference Standard
| CAS Number | Not assigned |
| Formula | As defined in the European Pharmacopoeia (structurally related indole alkaloid derivative of yohimbi |
| Molecular Weight | Approximately 340–360 g/mol (per EP definition) |
| Purity | As per EP reference standard specification |
| Storage | Store at 2–8 °C; protect from light and moisture |
Yohimbine EP Impurity A is a specified related substance of yohimbine arising from synthesis or degradation, structurally related to the parent indole alkaloid.
Used as a European Pharmacopoeia reference standard for impurity identification, quantification, and EP compliance testing of yohimbine API and formulations.
Evaluated using EP monograph HPLC methods, with identity confirmation by LC-MS and retention time comparison to the EP CRS.