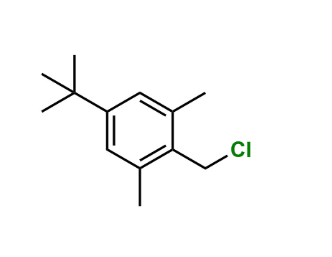
Reference Standard
| CAS Number | Not assigned |
| Formula | C₁₆H₂₄N₂O (approximate, related derivative) |
| Molecular Weight | ~252–260 g/mol |
| Purity | As per EP reference standard |
| Storage | Store refrigerated (2–8 °C) and protect from light |
EP Impurity B is a pharmacopoeial impurity of xylometazoline, a nasal decongestant, defined in the European Pharmacopoeia.
Used in EP compliance testing and impurity quantification for xylometazoline API.
HPLC/UPLC per EP monograph, LC-MS for identity confirmation.