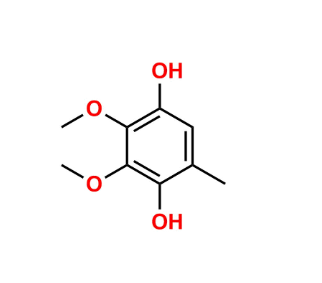
Reference Standard
| CAS Number | Not assigned |
| Formula | As defined in the European Pharmacopoeia (structurally related quinone derivative) |
| Molecular Weight | ~840–880 g/mol |
| Purity | As per EP reference standard specification |
| Storage | Store refrigerated (2–8 °C), protected from light |
EP Impurity A is a specified related substance of ubidecarenone described in the European Pharmacopoeia, arising from synthesis or degradation.
Used for EP compliance testing and impurity quantification in ubidecarenone API.
HPLC comparison to EP standard, LC-MS confirmation, system suitability testing.