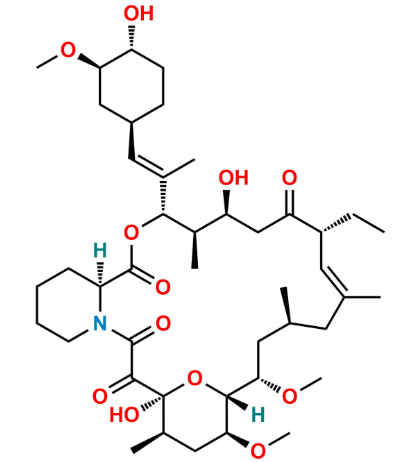
Reference Standard
| CAS Number | Not assigned |
| Formula | As defined in European Pharmacopoeia |
| Molecular Weight | Approximately 800 g/mol |
| Purity | As per EP reference standard |
| Storage | Store frozen or refrigerated as recommended by supplier |
Another specified EP impurity of tacrolimus representing a structurally modified macrolide derivative.
Used for regulatory impurity profiling and validation of analytical methods for tacrolimus.
Determined by HPLC/UPLC, LC-MS, NMR, and EP monograph-based methods.