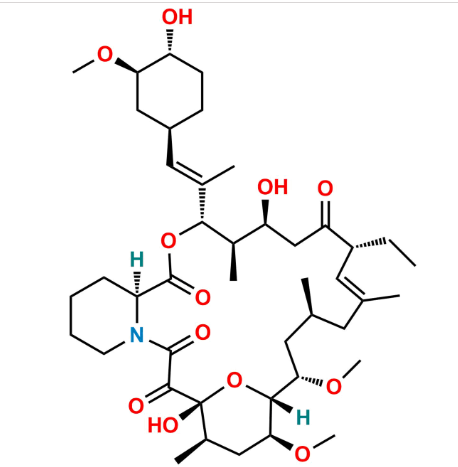
Reference Standard
| CAS Number | Not assigned |
| Formula | As defined in European Pharmacopoeia (related macrolide impurity) |
| Molecular Weight | Approximately 786–820 g/mol |
| Purity | As per EP reference standard requirements |
| Storage | Store at −20 °C to 2–8 °C; protect from light |
A specified European Pharmacopoeia impurity of tacrolimus, structurally related and formed during synthesis or degradation.
Used for EP compliance testing, impurity quantification, and batch release of tacrolimus.
HPLC comparison with EP standard, LC-MS confirmation, and system suitability testing.